The I-REIVAC network is piloted by a coordination team based at the Hôtel-Dieu Hospital in Paris. The network is currently governed by two bodies.
The network's coordination team
The coordination team, located at the Hôtel Dieu Hospital in Paris, is led by Pr Odile Launay (Infectiologist, PU-PH, Hôpital Cochin, AP-HP) and Pr Eric Tartour (Immunologist, PU-PH, Hôpital Européen Georges-Pompidou, HEGP) who joined the governance in 2023.
The team's legal representation is covered by Inserm and AP-HP, the trustees of the I-REIVAC network.
It includes the network coordinators, deputy coordinators, a secretary, a financial manager, project managers and support staff.
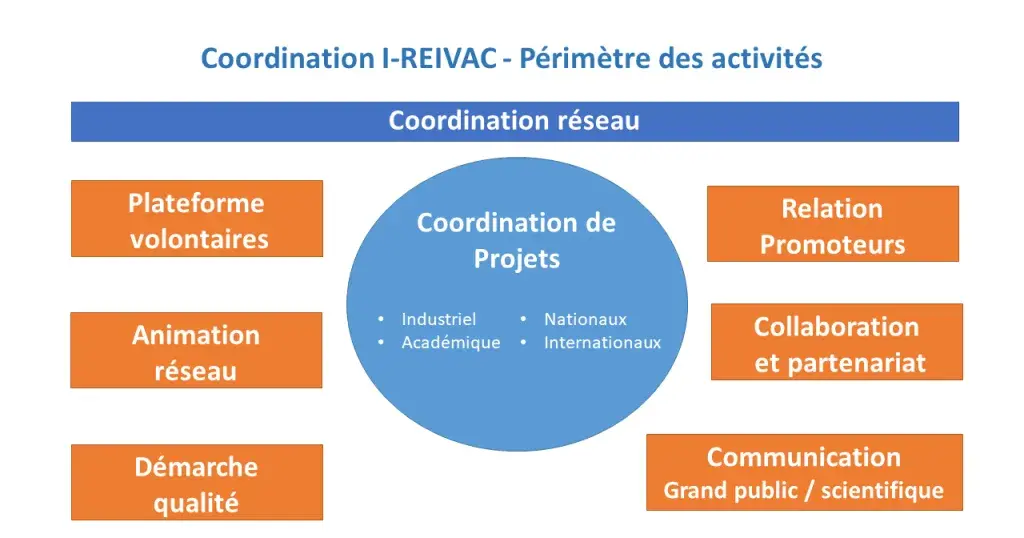
The coordination team is the network's "main entrance", and :
- Provides a key link to promoters and network members;
- Centralises information and ensures its dissemination to all members;
- Sets up and carries out projects;
- Facilitates and promotes a collective clinical research dynamic, by creating working groups, organising seminars, managing websites, sending out newsletters, etc.
- Sets up and ensures the collection of indicators used to monitor the network's activity;
- Prepares the network's annual activity report;
- Organises the communication on behalf of the network;
- Assures the network's valorisation;
- Provides a key interface with industrial and institutional partner organisations;
- Develops knowledge, and the sharing and transmission of expertise within the network ;
- Proposes strategic areas of work and development;
- Formulates recruitment strategies, in particular via the CoV-IREIVAC national online volunteer platform;
- Works to ensure the network's long-term viability, in particular by seeking financial support and building an economic model;
- On a day-to-day basis, handles the network's financial monitoring in liaison with the supervisory bodies.
The coordination team is the one-stop entry point for all network requests from all partners: researchers and institutional and industrial partners/promoters. In particular, it is responsible for coordinating, sharing information, and monitoring the activities carried out within the network, as well as preparing and monitoring the governance committees (Steering Committee and Scientific Advisory Board). Particular attention is paid to communication towards volunteers and the scientific community, including with webinars and symposia.
In this context, the coordination team implements the following activities:
- Ensures a regular link with all institutions/sponsors (academic and industrial) to discuss their scientific strategy and inform them about the network's activities, expertise and operational developments;
- Establishes and manages institutional partnerships (public and private) to contribute to the scientific and organisational development of the network;
- Supports the implementation of academic and industrial research projects and ensures the link between promoters and network players (clinic, CRB, laboratory, CMG/URC, ...)
- Animation of the I-REIVAC network and its components
- Administers and animates the national volunteer registry (CoV-IREIVAC);
- Maintains a quality assurance program on its activities
- Ensures effective communication to volunteers (Letters of information, webinars, newsletters, symposia)
Activities of the Coordination Team
Governance
The network's current governance is based on two bodies:
International Scientific Advisory Board
The network set up an independent scientific committee made up of a dozen French and European scientific experts in the field of vaccinology. The network's supervisory bodies and the private sector are not represented on this committee in order to maintain the scientific committee's independence.
The scientific committee's missions are to provide feedback on :
- the network's scientific strategy,
- the network's objectives and priorities ,
- the analysis of the network's strengths and weaknesses.
The independent scientific committee meets every 2 years, preferably face-to-face. Minutes are systematically drawn up and circulated to all members of the Scientific Committee. Exceptional meetings can be organised whenever necessary.
Steering committee
The Steering committee is made up of the coordinators, the two deputy coordinators, representatives of the network centres and external members. The steering committee is warrant of the network's smooth operation and achievement of its objectives.
The steering committee is responsible for:
- monitoring the network's activities,
- validating the scientific interest of studies contemplated by the network,
- deciding on the integration/exclusion of a centre, network member or partner,
- also establishing the network's priorities and objectives.
The steering committee meets online every 2 months. Minutes are systematically drawn up and circulated to all steering committee members. Exceptional meetings can be organised whenever necessary.
Members of the steering committee commit to respect the confidential nature of the information to which they access when submitting a project.
The steering committee is composed of:
- Odile Launay, infectiologist, University Professor at Paris University and Hospital Practitioner at Cochin Hospital, network coordinator and clinical research referent
- Eric Tartour, Professor of Immunology at the University of Paris and head of the biological immunology department at the Hôpital Européen Georges Pompidou, referent on markers of immune response and network coordinator
- Louise Lefrançois, coordinating team
- Florian Verrier, coordinating team
- Florence Galtier, medical manager of the Clinical Investigation Center of Montpellier University Hospital, co-referent on vaccination of special populations
- Philippe Vanhems, epidemiologist, University Professor and Hospital Practitioner at the Hospices Civils de Lyon, referent on the evaluation of vaccine efficacy and pharmaco-epidemiology
- Catherine Mutter, Physician Delegate at the Clinical Investigation Center of Strasbourg University Hospitals
- Paul Loubet, specialist in Infectious and Tropical Diseases at Nîmes University Hospital. Co-coordinator of the COV-POPART cohort and principal investigator for the COVI-COMPARE-P and ARN-combi trials.
- Anne Conrad, medical manager of the Clinical Research Center at Hôpital de la Croix-Rousse, Hospices Civils de Lyon
- Liem Binh Luong, assistant physician at CIC Cochin Pasteur, and infectiologist, Harvard University graduate in public health and École Normale Supérieure alumnus.
- Elisabeth Botelho-Nevers, an infectiologist at Saint-Etienne University Hospital, is in charge of the CIC's vaccinology axis. Coordinator of the COVI-COMPARE-J trial.
- Béatrice Parfait, Professor of Molecular Genetics at the University of Paris Cité and head of the Cochin Biological Resources Center
- Christian Dualé, physician delegate at the Centre d'Investigation Clinique at the CHU Clermont-Ferrand, referent on quality and ethical dimensions
- Régis Hankard, University Professor of Pediatrics at Université De Tours-CHU Tours, representative of the F-CRIN PEDSTART network
- Florence Flamein, Pediatric Neonatologist at the Clinical Investigation Center, representative of the F-CRIN PEDSTART network
- Karine Petitprez, Chargée de mission-department soutiens structurants à la recherche de l'ANRS Maladies infectieuses émergentes

